Cat.NO.:A357114 Purity:99%
Product Details of BV6
| CAS No. : | 1001600-56-1 |
| Formula : |
C70H96N10O8 |
| M.W : |
1205.57
|
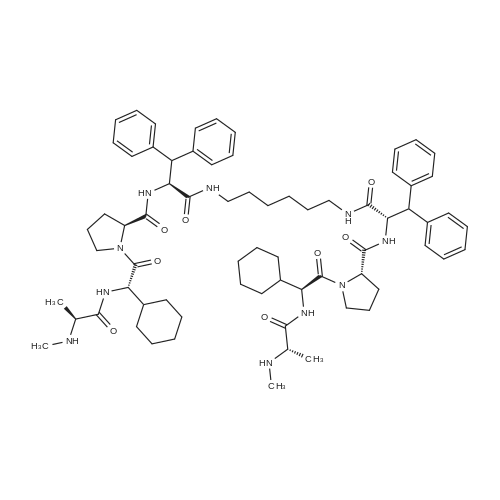
| SMILES Code : | O=C(NCCCCCCNC([C@@H](NC([C@H](CCC1)N1C([C@H](C2CCCCC2)NC([C@H](C)NC)=O)=O)=O)C(C3=CC=CC=C3)C4=CC=CC=C4)=O)[C@@H](NC([C@H](CCC5)N5C([C@H](C6CCCCC6)NC([C@H](C)NC)=O)=O)=O)C(C7=CC=CC=C7)C8=CC=CC=C8 |
| Synonyms : |
BV-6 free
|
| MDL No. : | MFCD28168021 |
Safety of BV6
| GHS Pictogram: |  |
| Signal Word: | Warning |
| Hazard Statements: | H302-H315-H319-H335 |
| Precautionary Statements: | P261-P305+P351+P338 |
Isoform Comparison
Biological Activity
In Vitro:
| Concentration | Treated Time | Description | References |
| HT29 cells | 2 μM | 1, 2, 4 hours | Induction of necroptosis in HT29 cells, stimulating RIP1 ubiquitination within the necrosome | PMC5260504 |
| GBM9 cells | 0.6 µM | 10 days | BV6 stimulates morphological changes in GBM9 cells and increases GFAP expression, indicating that BV6 induces astrocytic differentiation of GBM9 cells. | PMC3978303 |
| A172 glioblastoma cells | 2 μM | 120 hours | BV6 enhances TMZ-induced apoptosis, and upregulation of IFNβ is a critical event in BV6/TMZ-induced apoptosis. | PMC4650438 |
| T98G glioblastoma cells | 4 μM | 120 hours | BV6 enhances TMZ-induced apoptosis, and upregulation of IFNβ is a critical event in BV6/TMZ-induced apoptosis. | PMC4650438 |
| L929 cells | 1 µM | 2 hours | BV6 treatment greatly sensitized L929 cells to TNF-induced necrosis, but not to necrotic death induced by anti-Fas, poly(I:C), or oxidative stress. | PMC3131911 |
| acute lymphoblastic leukemia cells | 300 nM | 24 hours | induces necroptotic cell death | PMC5260489 |
| mouse embryonic fibroblasts | 5 μM | 24 hours | protects cells from BV6/Dexa-induced cell death | PMC5260489 |
| HCC193 | 1 μM | 24 hours | BV6 significantly enhanced the radiosensitivity of HCC193 cells, DER=1.38 (p<0.05). | PMC3196825 |
| FADD-deficient Jurkat T cells | 1 µM | 24 hours | BV6 treatment sensitized FADD-deficient Jurkat T cells to TNF-induced necrosis. | PMC3131911 |
| T98G cells | 2.5 µM | 24 hours | BV6 at non-toxic concentrations triggers cell elongation, migration, and invasion, and causes profound depletion of cIAP1 protein. | PMC3615728 |
| U87MG cells | 2.5 µM | 24 hours | BV6 at non-toxic concentrations triggers cell migration and invasion, and causes degradation of cIAP1 and XIAP proteins. | PMC3615728 |
| LN229 cells | 2.5 µM | 24 hours | BV6 at non-toxic concentrations triggers cell migration and invasion, and causes degradation of cIAP1 and XIAP proteins. | PMC3615728 |
| GBM1 cells | 1 µM | 24 hours | BV6 at non-toxic concentrations triggers cell elongation, migration, and invasion, and causes profound depletion of cIAP1 protein. | PMC3615728 |
| GBM2 cells | 0.8 µM | 24 hours | BV6 at non-toxic concentrations triggers cell elongation, migration, and invasion, and causes profound depletion of cIAP1 protein. | PMC3615728 |
| AsPc-1 | 5 µM | 48 h | To investigate whether BV6 combined with 2′3′-cGAMP can induce necroptosis in apoptosis-deficient pancreatic cancer cells, the results showed that BV6 combined with 2′3′-cGAMP significantly increased cell death, and this death could be reduced by Nec-1s inhibition of RIPK1, indicating RIPK1-dependent necroptotic cell death. | PMC8405653 |
| BxPc-3 | 5 µM | 48 h | To investigate whether BV6 combined with 2′3′-cGAMP can induce necroptosis in apoptosis-deficient pancreatic cancer cells, the results showed that BV6 combined with 2′3′-cGAMP significantly increased cell death, and this death could be reduced by Nec-1s inhibition of RIPK1, indicating RIPK1-dependent necroptotic cell death. | PMC8405653 |
| Capan-1 | 5 µM | 48 h | To investigate whether BV6 combined with 2′3′-cGAMP can induce necroptosis in apoptosis-deficient pancreatic cancer cells, the results showed that BV6 combined with 2′3′-cGAMP significantly increased cell death, and this death could be reduced by Nec-1s inhibition of RIPK1, indicating RIPK1-dependent necroptotic cell death. | PMC8405653 |
| DanG | 5 µM | 48 h | To investigate whether BV6 combined with 2′3′-cGAMP can induce necroptosis in apoptosis-deficient pancreatic cancer cells, the results showed that BV6 combined with 2′3′-cGAMP significantly increased cell death, and this death could be reduced by Nec-1s inhibition of RIPK1, indicating RIPK1-dependent necroptotic cell death. | PMC8405653 |
| H460 | 5 μM | 48 hours | BV6 significantly enhanced the radiosensitivity of H460 cells, DER=1.42 (p<0.05). | PMC3196825 |
| UoCB6 | 66.1 nM | 48 hours | BV6 induced cell death, primarily through TNF-α-dependent apoptosis | PMC4816168 |
| REH | 251.1 nM | 48 hours | BV6 induced cell death, primarily through TNF-α-dependent apoptosis | PMC4816168 |
| GBM10 cells | 1 µM | 7 days | BV6 stimulates morphological changes in GBM10 cells and increases GFAP expression, indicating that BV6 induces astrocytic differentiation of GBM10 cells. | PMC3978303 |
| Non-malignant neural stem cells (NSCs) | 1 µM | 7 days | BV6 does not alter cell morphology, differentiation, or expression of stemness markers in NSCs. | PMC3978303 |
| A172 glioblastoma cells | 5 μM | 72 hours | To investigate the mechanism of BV6-induced apoptosis, it was found that BV6 upregulates DR5 expression in an NF-κB-dependent manner, thereby promoting apoptosis. | PMC3847333 |
| MDA-MB-231 breast carcinoma cells | 50 nM | 72 hours | To investigate the mechanism of BV6-induced apoptosis, it was found that BV6 induces apoptosis through the TNFα/TNFR1 autocrine/paracrine signaling pathway. | PMC3847333 |
| MDA-MB-231 cells | 50 nM | 72 hours | To evaluate BV6-induced cell death, results showed that IRF1 knockdown significantly reduced BV6-induced cell death. | PMC4454156 |
| T24 cells | 100 nM | 72 hours | To evaluate BV6-induced cell death, results showed that IRF1 knockdown significantly reduced BV6-induced cell death. | PMC4454156 |
| SK-N-AS cells | 50 nM | 72 hours | To evaluate BV6-induced cell death, results showed that IRF1 knockdown significantly reduced BV6-induced cell death. | PMC4454156 |
| Kym1 cells | 100 nM | 72 hours | To evaluate BV6-induced cell death, results showed that IRF1 knockdown significantly reduced BV6-induced cell death. | PMC4454156 |
| H1299 cells | 1 μM | various time periods | To evaluate the effect of BV6 on cIAP1 and cIAP2 protein levels, results showed that cIAP2 degradation was protected in H1299 cells | PMC4532773 |
In Vivo:
| Administration | Dosage | Frequency | Description | References |
| WT and Ccr2−/− mice | acute sclerosing cholangitis model | intrabiliary injection | single intrabiliary injection | single injection, continued for 5 days | inhibition of monocyte/macrophage recruitment by genetic or pharmacological depletion of CCR2 reduces biliary injury and fibrosis | PMC6098983 |
| Athymic Nude mice | Orthotopic and subcutaneous GBM models | Injection | 0.6 mM | Single injection, observed until clinical symptoms appeared | BV6 treatment reduces clonogenicity and tumorigenicity of GBM CSLCs in vivo and significantly increases the survival of mice. | PMC3978303 |
| Nude mice | xenograft tumor model | intraperitoneal injection | 10 mg/kg | every 4 days, total four treatments | To evaluate the tumor suppressive effect of BV6, results showed that USP11 downregulation enhanced the antitumor activity of BV6 and TRAIL | PMC4532773 |
| NOD/SCID mice | huALL model | intraperitoneal injections | 10 mg/kg | twice per week for 2 weeks | BV6 significantly reduced leukemia load and prolonged leukemia-free survival, and this effect was dependent on TNF-α | PMC4816168 |
| Chicken embryos | Chicken chorioallantoic membrane model | Implantation on the chicken chorioallantoic membrane | 2.5 µM | Single administration, lasting 4 days | Pre-treatment with BV6 significantly increased the infiltrative growth of GBM cells, indicating that BV6 enhances the invasiveness of GBM cells in vivo. | PMC3615728 |
Protocol
| Bio Calculators | ||||
| Preparing Stock Solutions |  |
1mg | 5mg | 10mg |
|
1 mM 5 mM 10 mM |
0.83mL 0.17mL 0.08mL |
4.15mL 0.83mL 0.41mL |
8.29mL 1.66mL 0.83mL |
|
| Dissolving Methods |
Please choose the appropriate dissolution scheme according to your animal administration guide.For the following dissolution schemes, clear stock solution should be prepared according to in vitro experiments, and then cosolvent should be added in turn:
in order to ensure the reliability of the experimental results, the clarified stock solution can be properly preserved according to the storage conditions; The working fluid for in vivo experiment is recommended to be prepared now and used on the same day; The percentage shown in front of the following solvent refers to the volume ratio of the solvent in the final solution; If precipitation or precipitation occurs in the preparation process, it can be assisted by heating and/or ultrasound.
|
|||


Reviews
There are no reviews yet.