Cat.NO.:A560920 Purity:97%
Product Details of [ 1005508-80-4 ]
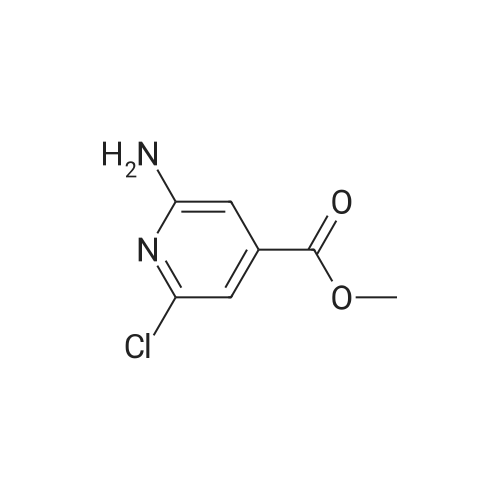
| CAS No. : | 1005508-80-4 |
| Formula : |
C7H7ClN2O2 |
| M.W : |
186.60
|
| SMILES Code : | O=C(OC)C1=CC(Cl)=NC(N)=C1 |
| MDL No. : | MFCD18447558 |
| InChI Key : | MYBUXYUDDLFCPJ-UHFFFAOYSA-N |
| Pubchem ID : | 55251669 |
Safety of [ 1005508-80-4 ]
| GHS Pictogram: |  |
| Signal Word: | Danger |
| Hazard Statements: | H301-H311-H331 |
| Precautionary Statements: | P261-P264-P270-P271-P280-P302+P352-P304+P340-P310-P330-P361-P403+P233-P405-P501 |
| Class: | 6.1 |
| UN#: | 2811 |
| Packing Group: | Ⅲ |
Application In Synthesis of [ 1005508-80-4 ]
* All experimental methods are cited from the reference, please refer to the original source for details. We do not guarantee the accuracy of the content in the reference.
- Downstream synthetic route of [ 1005508-80-4 ]
[ 1005508-80-4 ] Synthesis Path-Downstream 1~35
| Yield | Reaction Conditions | Operation in experiment |
|---|---|---|
| 95% | In propan-1-ol; for 20h;Heating / reflux; | Methyl 2-amino-6-chloro-isonicotinate (9.33 g, 50.0 mmol) was suspended in n-propanol. 1-Bromo-2,2-dimethoxypropan (20.26 ml, 150 mmol) was added dropwise. After stirring at reflux for 20 h, n-propanol was removed in vacuo and the residue was recrystallized from diisopropyl ether. 14.55 g (95%) of the title compound were obtained as pale brown solid.1H-NMR (200 MHz, d6-DMSO): δ = 8.29 (s, 1 H), 8.25 (s, 1 H), 7.83 (s, 1 H), 6.14 (br s), 3.95 (s, 3H), 2.53 (s, 3H). |
References: [1]Patent: WO2011/159864,2011,A1 .
| Yield | Reaction Conditions | Operation in experiment |
|---|---|---|
| 40% | With tert.-butylnitrite; diiodomethane; iodine; for 5.08333h; | Iodine (2.84 g, 1 1 .2 mmol ) was the added to the reaction3 mixture and stirred for 5 h. The reaction mixture was diluted with ethyl acetate, washed with a mixture of (NaHC03) (100 ml ) and 20% sodium thiosulfate (25.0 mL), and dried. (Na2S04). Concentration of ethyl acetate solution gave an oil, which was purified by silica gel column chromatography (hexane / ethyl acetate, 7 / 3) Product containing fractions were collected and concentrated to the give 25 as a light yellow solid. Yield 1.35 g (40%). MS: 297.9 (M+H f |
References: [1]Patent: WO2011/159864,2011,A1 .
References: [1]Patent: WO2011/159864,2011,A1 .
References: [1]Patent: WO2011/159864,2011,A1 .
References: [1]Patent: WO2011/159864,2011,A1 .
References: [1]Patent: WO2011/159864,2011,A1 .
References: [1]Patent: WO2011/159864,2011,A1 .
References: [1]Patent: WO2011/159864,2011,A1 .
| Yield | Reaction Conditions | Operation in experiment |
|---|---|---|
| 71% | Compound 24. El CI gas was passed through a solution o i -ani i no-6-ch loropyri dine -4- carboxylic acid2 in methanol (50.0 mL) until saturation and the reaction mixture was stirred for 12 h. Methanol ic solution was concentrated and the solid obtained was neutralized with a solution of sodium bicarbonate. The mixture was extracted with ethyl acetate, washed with water and dried (Na2S04). Ethyl acetate solution was concentrated to the methyl ester 14 as light brown solid. Yield 2.1 g (71 %). MS: 187.1 (M+H)+ |
References: [1]Patent: WO2015/118057,2015,A1 .
- 15
 [ 1005508-80-4 ]
[ 1005508-80-4 ]
- N-(2-chloro-6-fluoro-4-pyridyl)-3-fluoro-1-methyl-4-[[(1R)-2,2,2-trifluoro-1-methylethyl]sulfamoyl]pyrrole-2-carboxamide [ No CAS ]
References: [1]Patent: WO2015/118057,2015,A1 .
| Yield | Reaction Conditions | Operation in experiment |
|---|---|---|
| 2.2 g | With water; sodium hydroxide; In tetrahydrofuran; at 0 – 25℃; for 3h; | Trimethylsilyl)diazomethane (8.7 mL, 17.5 mmol) was added dropwise to a solution of 2-chloro- 6-[(2,4-dimethoxyphenyl)methylamino]pyridine-4-carboxylic acid (4.7 g, 14.56 mmol) in methanol (5 mL) and dichloromethane (15 mL) at 0C and stirred 25 minutes at 25C. The solution was concentrated resulting in methyl 2-chloro-6-[(2,4- dimethoxyphenyl)methylamino]pyridine-4-carboxylate (5.0g) as a yellow solid which was used as such. TFA (5 mL) was added to a solution of methyl 2-chloro-6-[(2,4- dimethoxyphenyl)methylamino]pyridine-4-carboxylate (5.0g, 14.9 mmol) in dichloromethane (20 mL) and stirred 30 minutes at 25C. The reaction mixture was concentrated. The resulting solid was triturated 30 minutes in methyl tert-butyl ether (40 mL). The solids were filtered off and washed with methyl tert-butyl ether (10 mL) resulting in methyl 2-amino-6-chloro-pyridine- 4-carboxylate (3.0 g) as a light yellow solid. Sodium hydroxide (3.0g, 75.0 mmol) was added to a solution of methyl 2-amino-6-chloro-pyridine-4-carboxylate (2.8 g, 15 mmol) in water (10 mL) and THF (40 mL) at 0C and stirred 3 hours at 25C. The reaction mixture was cooled to 0C and acidified with HC1 (7 mL). The brown solid was filtered off, washed with water (30 mL) and THF (10 mL) and was lyophilized, resulting in 2-amino-6-chloro-pyridine-4-carboxylic acid (2.2g). 1H NMR (400MHz, DMSO-d6) δ = 6.87 (s, 1H), 6.80 (s, 1H), 6.71 (br. s., 2H). 2-amino- 6-chloro-pyridine-4-carboxylic acid (1.2g, 6.95 mmol) was dissolved in toluene (10 mL). Then triethylamine (2.97 g, 8.35 mmol), benzyl alcohol (3.01g, 27.82 mmol) and diphenylphosphoryl azide (2.97g, 8.35 mmol) were added and heated at 110C during 16 hours. The reaction mixture was cooled. EtOAc (100 mL) was added and washed with NaHC03 (sat.aq. 60 mL). The mixture was filtered and the solids washed with EtOAc (30 mL) the organic layer was separated from the filtrate. The water layer was extracted with EtOAc (3 x 30mL) The organic layers were combined and washed with brine (70 mL), dried over Na2S04, filtered and concentrated under reduce pressure resulting in a brown oil. The oil was purified by column chromatography (Si02, Petroleum ether/Ethyl acetate=0 to 50% resulting in benzyl N-(2-amino-6-chloro-4- pyridyl)carbamate as a colorless oil. Sodium nitrite (894 mg, 12.96 mmol) was added to a solution of benzyl N-(2-amino-6-chloro-4-pyridyl)carbamate (360 mg, 1.30 mmol) in pyridine hydrofluoride (1 mL) at 0C and stirred 2 hours at 25C. NaHC03 (100 mL sat.aq.) was added. The mixture was extracted with EtOAc (3 x 30 mL). The combined organic layers were washed with brine (70 mL), dried over Na2S04, filtered, and concentrated in vacuo. The obtained residue was purified by column chromatography (Si02, Ethyl acetate/Petroleum ether=0 to 30%, TLC pentane:EtOAc =3: 1, Rf=0.8) resulting in benzyl N-(2-chloro-6-fluoro-4-pyridyl)carbamate (200 mg) as a colorless oil. Platinumdioxide (10 mg) was added under nitrogen to a solution of benzyl N-(2-chloro-6-fluoro-4-pyridyl)carbamate in methanol (20 mL) and the mixture was stirred under hydrogen atmospohere for 16 hours at 25C. The mixture was filtered and the filtrate was concentrated under reduced pressure. The residue was purified by prep-HPLC (Column: Phenomenex Gemini 150*25mm* 10um. Condition: 0.05%ammonia-ACN). The obtained solid was dried resulting in 2-chloro-6-fluoro-pyridin-4-amine (33 mg) as a white solid. 1H NMR (400MHz, DMSO-d6) δ = 6.83 (br. s., 2H), 6.46 (t, J=1.6 Hz, 1H), 6.06 (d, J=1.6 Hz, 1H). |
References: [1]Patent: WO2015/118057,2015,A1 .
References: [1]Patent: WO2015/118057,2015,A1 .
References: [1]Patent: WO2015/118057,2015,A1 .
- 20
- 2-chloro-6-[(2,4-dimethoxyphenyl)methylamino]pyridine-4-carboxylic acid [ No CAS ]

 [ 1005508-80-4 ]
[ 1005508-80-4 ]
References: [1]Patent: WO2015/118057,2015,A1 .
- 21
- methyl 2-chloro-6-[(2,4-dimethoxyphenyl)methylamino]pyridine-4-carboxylate [ No CAS ]

 [ 1005508-80-4 ]
[ 1005508-80-4 ]
| Yield | Reaction Conditions | Operation in experiment |
|---|---|---|
| 3 g | With triethylamine; In dichloromethane; at 25℃; for 0.5h; | Trimethylsilyl)diazomethane (8.7 mL, 17.5 mmol) was added dropwise to a solution of 2-chloro- 6-[(2,4-dimethoxyphenyl)methylamino]pyridine-4-carboxylic acid (4.7 g, 14.56 mmol) in methanol (5 mL) and dichloromethane (15 mL) at 0C and stirred 25 minutes at 25C. The solution was concentrated resulting in methyl 2-chloro-6-[(2,4- dimethoxyphenyl)methylamino]pyridine-4-carboxylate (5.0g) as a yellow solid which was used as such. TFA (5 mL) was added to a solution of methyl 2-chloro-6-[(2,4- dimethoxyphenyl)methylamino]pyridine-4-carboxylate (5.0g, 14.9 mmol) in dichloromethane (20 mL) and stirred 30 minutes at 25C. The reaction mixture was concentrated. The resulting solid was triturated 30 minutes in methyl tert-butyl ether (40 mL). The solids were filtered off and washed with methyl tert-butyl ether (10 mL) resulting in methyl 2-amino-6-chloro-pyridine- 4-carboxylate (3.0 g) as a light yellow solid. Sodium hydroxide (3.0g, 75.0 mmol) was added to a solution of methyl 2-amino-6-chloro-pyridine-4-carboxylate (2.8 g, 15 mmol) in water (10 mL) and THF (40 mL) at 0C and stirred 3 hours at 25C. The reaction mixture was cooled to 0C and acidified with HC1 (7 mL). The brown solid was filtered off, washed with water (30 mL) and THF (10 mL) and was lyophilized, resulting in 2-amino-6-chloro-pyridine-4-carboxylic acid (2.2g). 1H NMR (400MHz, DMSO-d6) δ = 6.87 (s, 1H), 6.80 (s, 1H), 6.71 (br. s., 2H). 2-amino- 6-chloro-pyridine-4-carboxylic acid (1.2g, 6.95 mmol) was dissolved in toluene (10 mL). Then triethylamine (2.97 g, 8.35 mmol), benzyl alcohol (3.01g, 27.82 mmol) and diphenylphosphoryl azide (2.97g, 8.35 mmol) were added and heated at 110C during 16 hours. The reaction mixture was cooled. EtOAc (100 mL) was added and washed with NaHC03 (sat.aq. 60 mL). The mixture was filtered and the solids washed with EtOAc (30 mL) the organic layer was separated from the filtrate. The water layer was extracted with EtOAc (3 x 30mL) The organic layers were combined and washed with brine (70 mL), dried over Na2S04, filtered and concentrated under reduce pressure resulting in a brown oil. The oil was purified by column chromatography (Si02, Petroleum ether/Ethyl acetate=0 to 50% resulting in benzyl N-(2-amino-6-chloro-4- pyridyl)carbamate as a colorless oil. Sodium nitrite (894 mg, 12.96 mmol) was added to a solution of benzyl N-(2-amino-6-chloro-4-pyridyl)carbamate (360 mg, 1.30 mmol) in pyridine hydrofluoride (1 mL) at 0C and stirred 2 hours at 25C. NaHC03 (100 mL sat.aq.) was added. The mixture was extracted with EtOAc (3 x 30 mL). The combined organic layers were washed with brine (70 mL), dried over Na2S04, filtered, and concentrated in vacuo. The obtained residue was purified by column chromatography (Si02, Ethyl acetate/Petroleum ether=0 to 30%, TLC pentane:EtOAc =3: 1, Rf=0.8) resulting in benzyl N-(2-chloro-6-fluoro-4-pyridyl)carbamate (200 mg) as a colorless oil. Platinumdioxide (10 mg) was added under nitrogen to a solution of benzyl N-(2-chloro-6-fluoro-4-pyridyl)carbamate in methanol (20 mL) and the mixture was stirred under hydrogen atmospohere for 16 hours at 25C. The mixture was filtered and the filtrate was concentrated under reduced pressure. The residue was purified by prep-HPLC (Column: Phenomenex Gemini 150*25mm* 10um. Condition: 0.05%ammonia-ACN). The obtained solid was dried resulting in 2-chloro-6-fluoro-pyridin-4-amine (33 mg) as a white solid. 1H NMR (400MHz, DMSO-d6) δ = 6.83 (br. s., 2H), 6.46 (t, J=1.6 Hz, 1H), 6.06 (d, J=1.6 Hz, 1H). |
- 22
 [ 1005508-80-4 ]
[ 1005508-80-4 ]
- 6-chloro-3-(3-(4-chloro-3,5-dimethylphenoxy)propyl)-7-(1,3,5-trimethyl-1H-pyrazol-4-yl)-1H-indole-2-carbonyl chloride [ No CAS ]

- methyl 2-chloro-6-(6-chloro-3-(3-(4-chloro-3,5-dimethylphenoxy)propyl)-7-(1,3,5-trimethyl-1H-pyrazol-4-yl)-1H-indole-2-carboxamido)isonicotinate [ No CAS ]
| Yield | Reaction Conditions | Operation in experiment |
|---|---|---|
| 100 mg | A solution of 6-chloro-3-(3-(4-chloro-3,5-dimethylphenoxy)propyl)-7-(l,3,5- trimethyl-lH-pyrazol-4-yl)-lH-indole-2-carbonyl chloride (100 mg, 0.19 mmol), DMAP (47 mg, 0.39 mmol), Pyridine (31 μ^, 0.39 mmol) in DCM (3.8 mL) was stirred at rt for 10 min, and <strong>[1005508-80-4]methyl 2-amino-6-chloroisonicotinate</strong> (40 mg, 0.21 mmol) was aded. The resulting mixture was stirred at rt for 15 h then concentrated. The residue was purified by silica gel flash chromatography (ISCO, 0-60% EtO Ac/Hex gradient) to give the title compound (100 mg, 0.149 mmol). MS (ES) 667.9 (M+H). |
- 23
 [ 1005508-80-4 ]
[ 1005508-80-4 ]
 [ 4637-24-5 ]
[ 4637-24-5 ]
- methyl 2-chloro-6-(N’-hydroxyimidamido)pyridine-4-carboxylate [ No CAS ]
| Yield | Reaction Conditions | Operation in experiment |
|---|---|---|
| Step l : A solution of <strong>[1005508-80-4]methyl 2-amino-6-chloropyridine-4-carboxylate</strong> (CAS 1005508-80-4, 10 g, 53.6 mmol) and DMF-DMA (7.18 mL, 53.6 mmol) was heated to 70 C for 2 h. More DMF- DMA (7.18 mL, 53.6 mmol) was added and the reaction was heated for a further 4h. The reaction was cooled to 50 C and hydroxylamine hydrochloride (3.72 g, 53.6 mmol) was added. The reaction was stirred at 50 C for 2 h. The reaction was concentrated in vacuo and the resulting residue was triturated with EtOH. The solid was filtered and dried under vacuum to afford methyl 2-chloro-6-(N’-hydroxyimidamido)pyridine-4-carboxylate. 1H NMR (400 MHz, DMSO- 6) δ ppm 3.32 (s, 3 H), 7.22 – 7.26 (m, 1 H), 7.57- 7.61 (m, 1 H) 7.69 – 7.74 (m, 1 H), 10.00 – 10.04 (m, 1 H), 10.40 (s, 1 H) MS ES+: 230 |
- 25
 [ 1005508-80-4 ]
[ 1005508-80-4 ]
- 5-{7-amino-[1,2,4]triazolo[1,5-a]pyridin-5-yl}-6-methylpyridine-2-carbonitrile [ No CAS ]
References: [1]Patent: WO2016/148306,2016,A1 .
- 26
 [ 1005508-80-4 ]
[ 1005508-80-4 ]
- N-[5-(4-cyano-2-methylphenyl)-[1,2,4]triazolo[1,5-a]pyridin-7-yl]-2-hydroxyacetamide [ No CAS ]
References: [1]Patent: WO2016/148306,2016,A1 .
- 27
 [ 1005508-80-4 ]
[ 1005508-80-4 ]
- N-[5-(4-cyano-2-methylphenyl)-[1,2,4]triazolo[1,5-a]pyridin-7-yl]-3,3,3-trifluoro-2-hydroxypropanamide [ No CAS ]
References: [1]Patent: WO2016/148306,2016,A1 .
- 28
 [ 1005508-80-4 ]
[ 1005508-80-4 ]
- N-[5-(4-cyano-2-methylphenyl)-[1,2,4]triazolo[1,5-a]pyridin-7-yl]-2-hydroxy-2-methylpropanamide [ No CAS ]
References: [1]Patent: WO2016/148306,2016,A1









Reviews
There are no reviews yet.