Cat.NO.:A266807 Purity:97% HPLC
Product Details of [ 100331-89-3 ]
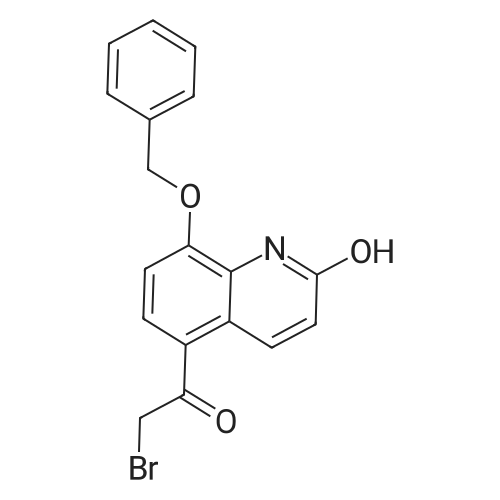
| CAS No. : | 100331-89-3 |
| Formula : |
C18H14BrNO3 |
| M.W : |
372.21
|
| SMILES Code : | BrCC(C1=C2C=CC(O)=NC2=C(OCC3=CC=CC=C3)C=C1)=O |
| MDL No. : | MFCD09025318 |
| InChI Key : | RVHSDLUBNZBRMH-UHFFFAOYSA-N |
| Pubchem ID : | 11667867 |
Safety of [ 100331-89-3 ]
| GHS Pictogram: |   |
| Signal Word: | Danger |
| Hazard Statements: | H302-H314 |
| Precautionary Statements: | P260-P264-P270-P280-P301+P330+P331-P303+P361+P353-P304+P340-P305+P351+P338-P310-P363-P405-P501 |
| Class: | 8 |
| UN#: | 3261 |
| Packing Group: | Ⅱ |
Application In Synthesis of [ 100331-89-3 ]
* All experimental methods are cited from the reference, please refer to the original source for details. We do not guarantee the accuracy of the content in the reference.
- Downstream synthetic route of [ 100331-89-3 ]
[ 100331-89-3 ] Synthesis Path-Downstream 1~6
| Yield | Reaction Conditions | Operation in experiment |
|---|---|---|
| 99% | With (S,S)-1,2-diphenyl-1,2-diaminoethane; hydrogen; potassium hydroxide; In isopropyl alcohol; at 25℃; under 3800.26 Torr;Autoclave; Inert atmosphere; | 1 kg of 8-benzyloxy-5-(2-bromoacetyl)-2-hydroxyquinoline was added to the autoclave.Add 10L of isopropanol,Then add 0.1g RuCl2-[(S)-BINAP](DMF)n,0.1 g of (S,S)-1,2-diphenylethylenediamine and 0.15 g of potassium hydroxide,Nitrogen replacement, hydrogen gas 5atm, temperature control 25 C reaction; after the reaction is completed,Nitrogen replacement, pressure filtration, concentration and crystallization of the filtrate, filtration,995g of 8-benzyloxy-5-(R-2-bromo-1-hydroxyethyl)-1H-quinolin-2-one with a chiral purity of 97.5%.The yield was 99%. |
| 95% | 10068] To a mixture of tetrahydroffiran (1400 ml) and Bromo compound (100 g), R-methyl CBS (7.5 g) was slowly added at 25-30 C. under nitrogen atmosphere and cooled to 0-5 C., followed by the addition of Boron Dimethyl Sulfide complex (24.3 g) at 0-10 C. The reaction mass was stirred for 1-2 hr at 0-10 C. The progress of the reaction was monitored by HPLC. After the completionthe reaction, methanol (100 ml) was slowly added to the reaction mass for 30 minutes and stirred for 15 minutes0-10 C. The contents were distilled under vacuum attemperature of 40-45 C. and cooled to 25-30 C. The cooled reaction mixture was slowly added to hydrochloric acid solution at 25-30 C. and stirred for 1-2 hours at the same temperature. The resulted solid was filtered, washed with water (300 ml) and dried at 65-70 C. for 8 hours. % Yield:95% | |
| 85.5% | With dimethylsulfide borane complex; (1R,2S)-1-Amino-2-indanol; In tetrahydrofuran; at 20 – 25℃; for 3.5h;Inert atmosphere; | [0160] 0.12 g (0.79 mmol) of (1R,2S)-(+)-1-amino-2-indenol was placed in a 500 mL three-necked flask. 50 mL of THF was added and stirred at 20-25 C. 1.1 ml (11.6 mmol) of borane dimethyl sulfide was added and stirred for 20 min. At the same time, 37.2g (100.0 mmol) of 8-benzyloxy-5-(2-bromoacetyl)-(1H)-quinolin-2-one and 500 ml of THF solution and 8.1 ml (86.3 mmol) of borane dimethyl sulfide and 120 ml of THF solution were added dropwise at 20-25 C for 3 hours. The reaction was kept at the temperature of 20-25 C and reacted for 30 min under nitrogen protection. TLC analysis showed that the reaction was complete. With external ice bath cooling (< 10 C), 50 mL of methanol was added dropwise, and the reaction was kept in the ice bath (<10 C) and stirred for 10 minutes after dropwise addition. The solvent was removed below 45 C under reduced pressure by a water pump. 200 mL of water and 200 ml of ethyl acetate were added and stirred at room temperature until bubbles were generated, stirred at room temperature for 5 minutes, and transferred into a separatory funnel. The organic layer was separated out. The water layer was further extracted with ethyl acetate (50 ml3 times). The organic layers were combined and dried over anhydrous magnesium sulfate. The desiccant was removed by filtration, and the solvent was removed below 45 C under reduced pressure by a water pump to obtain a crude product. The crude product was thermally dissolved with 400 mL acetonitrile. 3 g of activated carbon was added. Reflux was performed for 5 minutes, filtration was performed when it was hot, and crystals were precipitated by cooling. The solid was filtered and collected to obtain 31.8 g of 8-benzyloxy-5-[(R)-2-bromo-1-hydroxyethyl]-1H-quinolin-2-one with a yield of 85.5%. |
| 81% | EXAMPLE 2; Synthesis of N-{2-[4-(3-phenyl-4-methoxyphenyl)aminophenyl]ethyl}-(R)-2-hydroxy-2-(8-benzyloxy-2(1H)-quinolinon-5-yl)ethylamine (PP); a. Synthesis of 5-(2-bromo-(R)-1-hydroxy)ethyl-8-benzyloxy-2(1H)-quinolinone (FF); (R)-(+)-alpha,alpha-Diphenylprolinol (30.0 g, 117 mmol) and trimethylboroxine (11.1 ml, 78 mmol) were combined in toluene (300 ml) and stirred at room temperature for 30 minutes. The mixture was placed in a 150 C. oil bath and liquid was distilled off. Toluene was added in 20 ml aliquots, and distillation was continued for 4 hours. A total of 300 ml toluene was added. The mixture was finally cooled to room temperature. A 500 muL aliquot was evaporated to dryness, weighed (246 mg) to determine that the concentration of catalyst was 1.8 M. [0128] 5-(2-Bromo-1-oxy)ethyl-8-benzyloxy-2(1H)-quinolinone (R) (90.0 g, 243 mmol) was placed under nitrogen, tetrahydrofuran (900 mL) was added followed by the catalyst from above (1.8 M in toluene, 15 ml, 27 mmol). The suspension was cooled to -10+5 C. in an ice/isopropanol bath. Borane (1.0 M in THF, 294 ml, 294 mmol) was added over 4 hours. The reaction was stirred an additional 45 minutes at -10 C., then methanol (250 ml) was added slowly. The mixture was concentrated under vacuum. The residue was dissolved in boiling acetonitrile (1.3 l), filtered while hot and cooled to room temperature. The crystals were filtered, washed with acetonitrile and dried under vacuum to give 5-(2-bromo-(R)-1-hydroxy)ethyl-8-benzyloxy-2(1H)-quinolinone (FF) (72.5 g, 196 mmol, 81% yield, 95% ee, 95% pure by HPLC area ratio). | |
| 81% | With borane;(3aR)-1-methyl-3,3-diphenyl-tetrahydro-pyrrolo[1,2-c][1,3,2]oxazaborole; In tetrahydrofuran; toluene; at -10℃; for 4.75h; | (R)-(+)-alpha,alpha-Diphenylprolinol (30.0 g, 117 mmol) and trimethylboroxine (11.1 mL, 78 mmol) were combined in toluene (300 mL) and stirred at room temperature for 30 min. The mixture was placed in a 150 C. oil bath and liquid was distilled off. Toluene was added in 20 mL aliquots and distillation was continued for 4 h. A total of 300 mL toluene was added. The mixture was then cooled to room temperature. A 500 muL aliquot was evaporated to dryness and weighed (246 mg) to determine that the concentration of catalyst was 1.8M. [0523] 8-Benzyloxy 5-(2-bromoacetyl)-1H-quinolin-2-one (90.0 g, 243 mmol) was placed under nitrogen and tetrahydrofuran (900 mL) was added followed by the catalyst described above (1.8 M in toluene, 15 mL, 27 mmol). The suspension was cooled to -10+5 C. in an ice/isopropanol bath. Borane (1.0 M in THF, 294 mL, 294 mmol) was added over 4 h. The reaction was then stirred an additional 45 min at -10 C. and then methanol (250 mL) was added slowly. The mixture was concentrated under vacuum and the residue was dissolved in boiling acetonitrile (1.3 L), filtered while hot and then cooled to room temperature. The crystals were filtered, washed with acetonitrile and dried under vacuum to give the title compound (72.5 g, 196 mmol, 81% yield, 95% ee, 95% pure by HPLC). |
| 81% | (b) 8-Benzyloxy-5-((R)-2-bromo-1-hydroxyethyl)-1H-quinolin-2-one (R)-(+)-alpha,alpha-Diphenylprolinol (30.0 g, 117 mmol) and trimethylboroxine (11.1 ML, 78 mmol) were combined in toluene (300 ML) and stirred at room temperature for 30 min.The mixture was placed in a 150 C. oil bath and liquid was distilled off.toluene was added in 20 ML aliquots and distillation was continued for 4 h.A total of 300 ML toluene was added.The mixture was then cooled to room temperature.A 500 muL aliquot was evaporated to dryness and weighed (246 mg) to determine that the concentration of catalyst was 1.8M. 8-Benzyloxy 5-(2-bromoacetyl)-1H-quinolin-2-one (90.0 g, 243 mmol) was placed under nitrogen and tetrahydrofuran (900 ML) was added followed by the catalyst described above (1.8 M in toluene, 15 ML, 27 mmol).The suspension was cooled to -10+5 C. in an ice/isopropanol bath.Borane (1.0 M in THF, 294 ML, 294 mmol) was added over 4 h.The reaction was then stirred an additional 45 min at -10 C. and then methanol (250 ML) was added slowly.The mixture was concentrated under vacuum and the residue was dissolved in boiling acetonitrile (1.3 L), filtered while hot and then cooled to room temperature.The crystals were filtered, washed with acetonitrile and dried under vacuum to give the title compound (72.5 g, 196 mmol, 81% yield, 95% ee, 95% pure by HPLC). | |
| 81% | (R)-(+)-alpha,alpha-Diphenylprolinol (30.0 g, 117 mmol) and trimethylboroxine (11.1 mL, 78 mmol) were combined in toluene (300 mL) and stirred at room temperature for 30 min. The mixture was placed in a 150 C. oil bath and liquid was distilled off. Toluene was added in 20 mL aliquots and distillation was continued for 4 h. A total of 300 mL toluene was added. The mixture was then cooled to room temperature. A 500 muL aliquot was evaporated to dryness and weighed (246 mg) to determine that the concentration of catalyst was 1.8 M. 8-Benzyloxy 5-(2-bromoacetyl)-1H-quinolin-2-one (90.0 g, 243 mmol) was placed under nitrogen and tetrahydrofuran (900 mL) was added followed by the catalyst described above (1.8 M in toluene, 15 mL, 27 mmol). The suspension was cooled to -10+-5 C. in an ice/isopropanol bath. Borane (1.0 M in THF, 294 mL, 294 mmol) was added over 4 h. The reaction was then stirred an additional 45 min at -10 C. and then methanol (250 mL) was added slowly. The mixture was concentrated under vacuum and the residue was dissolved in boiling acetonitrile (1.3 L), filtered while hot and then cooled to room temperature. The crystals were filtered, washed with acetonitrile and dried under vacuum to give the title compound (72.5 g, 196 mmol, 81% yield, 95% ee, 95% pure by HPLC). | |
| 81% | (7?)-(+)-a,a-Diphenylprolinol (30.0 g, 117 mmol) and trimethylboroxine (11.1 mL, 78 mmol) were combined in toluene (300 mL) and stirred at room temperature for 30 min. The mixture was placed in a 150 C oil bath and liquid was distilled off. Toluene was added in 20 mL aliquots and distillation was continued for 4 h. A total of 300 mL toluene was added. The mixture was then cooled to room temperature. A 500 uL aliquot was evaporated to dryness and weighed (246 mg) to determine that the concentration of catalyst was 1.8 M.8-Benzyloxy 5-(2-bromoacetyl)-l//-quinolin-2-one (90.0 g, 243 mmol) was placed under nitrogen and tetrahydrofuran (900 mL) was added followed by the catalyst described above (1.8 M in toluene, 15 mL, 27 mmol). The suspension was cooled to -10+-5 C in an ice/isopropanol bath. Borane (1.0 M in THF, 294 mL, 294 mmol) was added over 4 h. The reaction was then stirred an additional 45 min at -10 C and then methanol (250 mL) was added slowly. The mixture was concentrated under vacuum and the residue was dissolved in boiling acetonitrile (1.3 L), filtered while hot and then cooled to room temperature. The crystals were filtered, washed with acetonitrile and dried under vacuum to gjve the title compound (72.5 g, 196 mmol, 81% yield, 95% ee, 95% pure by HPLC). | |
| 81% | With borane;(3aR)-1-methyl-3,3-diphenyl-tetrahydro-pyrrolo[1,2-c][1,3,2]oxazaborole; In tetrahydrofuran; at -10 – 5℃; for 4.75h; | (R)-(+)-alpha,alpha-Diphenylprolinol (30.0 g, 117 mmol) and trimethylboroxine (11.1 mL, 78 mmol) were combined in toluene (300 mL) and stirred at room temperature for 30 minutes. The mixture was placed in a 150 C. oil bath and liquid was distilled off. Toluene was added in 20 mL aliquots, and distillation was continued for 4 hours. A total of 300 mL toluene was added. The mixture was finally cooled to room temperature. A 500 muL aliquot was evaporated to dryness, weighed (246 mg) to determine that the concentration of catalyst was 1.8 M. 5-(2-Bromo-1-oxy)ethyl-8-benzyloxy-2(1H)-quinolinone (R) (90.0 g, 243 mmol) was placed under nitrogen, tetrahydrofuran (900 mL) was added followed by the catalyst from above (1.8 M in toluene, 15 mL, 27 mmol). The suspension was cooled to -10+/-5 C. in an ice/isopropanol bath. Borane (1.0 M in THF, 294 mL, 294 mmol) was added over 4 hours. The reaction was stirred an additional 45 minutes at -10 C., then methanol (250 mL) was added slowly. The mixture was concentrated under vacuum. The residue was dissolved in boiling acetonitrile (1.3 L), filtered while hot and cooled to room temperature. The crystals were filtered, washed with acetonitrile and dried under reduced pressure to give 5-(2-bromo-(R)-1-hydroxy)ethyl-8-benzyloxy-2(1H)-quinolinone (FF) (72.5 g, 196 mmol, 81% yield, 95% ee, 95% pure by HPLC area ratio). |
| 81% | (R)-(+)-alpha,alpha-DIPHENYLPROLINOL (30.0 g, 117 mmol) and trimethylboroxine (11.1 mL, 78 mmol) were combined in toluene (300 mL) and stirred at room temperature for 30 min. The mixture was placed in a 150 C oil bath and liquid was distilled off. Toluene was added in 20 mL aliquots and distillation was continued for 4 h. A total of 300 mL toluene was added. The mixture was then cooled to room temperature. A 500 pL aliquot was evaporated to dryness and weighed (246 mg) to determine that the concentration of catalyst was 1. 8 M. 8-BENZYLOXY 5- (2-bromoacetyl)-lH-quinolin-2-one (90.0 g, 243 mmol) was placed under nitrogen and tetrahydrofuran (900 mL) was added followed by the catalyst described above (1. 8 M in toluene, 15 mL, 27 mmol). The suspension was cooled TO-1 C in an ice/isopropanol bath. Borane (1.0 M in THF, 294 mL, 294 mmol) was added over 4 h. The reaction was then stirred an additional 45 min at-10 C and then methanol (250 mL) was added slowly. The mixture was concentrated under vacuum and the residue was dissolved in boiling acetonitrile (1.3 L), filtered while hot and then cooled to room temperature. The crystals were filtered, washed with acetonitrile and dried under vacuum to give the title compound (72.5 g, 196 mmol, 81% yield, 95% EE, 95% pure by HPLC). | |
| 81% | Preparation 5 8-Benzyloxy-5-((R)-2-bromo-1-hydroxyethyl)-1H-quinolin-2-one (R)-(+)-alpha,alpha-Diphenylprolinol (30.0 g, 117 mmol) and trimethylboroxine (11.1 mL, 78 mmol) were combined in toluene (300 mL) and stirred at room temperature for 30 min. The mixture was placed in a 150 C. oil bath and liquid was distilled off. Toluene was added in 20 mL aliquots and distillation was continued for 4 h. A total of 300 mL toluene was added. The mixture was then cooled to room temperature. A 500 muL aliquot was evaporated to dryness and weighed (246 mg) to determine that the concentration of catalyst was 1.8 M. 8-Benzyloxy 5-(2-bromoacetyl)-1H-quinolin-2-one (90.0 g, 243 mmol) was placed under nitrogen and tetrahydrofuran (900 mL) was added followed by the catalyst described above (1.8 M in toluene, 15 mL, 27 mmol). The suspension was cooled to -10+-5 C. in an ice/isopropanol bath. Borane (1.0 M in THF, 294 mL, 294 mmol) was added over 4 h. The reaction was then stirred an additional 45 min at -10 C. and then methanol (250 mL) was added slowly. The mixture was concentrated under vacuum and the residue was dissolved in boiling acetonitrile (1.3 L), filtered while hot and then cooled to room temperature. The crystals were filtered, washed with acetonitrile and dried under vacuum to give the title compound (72.5 g, 196 mmol, 81% yield, 95% ee, 95% pure by HPLC). | |
| 76% | Preparation intermediate 4(R)-8-(Benzyloxy)-5-(2-bromo-l-hydroxyethyl)quinolin-2(l//)-one8-(Benzyloxy)-5-(2-bromoacetyl)quinolin-2(lH)-one (26.0 g, 69.9 mmol) and (R)-3,3-diphenyl-l -methyltetrahydro-3H-pyrrolo[l ,2- c][ l ,3,2]oxazaborole (21.3 g, 76.8 mmol) were azeotroped with toluene (x 3) then suspended in anhydrous THF (400 mL) under an atmosphere of nitrogen. The suspension was cooled to -20C (external temperature) and borane dimethyl sulfide complex solution (45.4 mL, 90.8 mmol, 2.0 M solution in THF) was added by syringe pump over 3 hours. After complete addition the reaction mixture was stirred for an hour before quenching with methanol (25 mL). The reaction was warmed to T over 20 minutes. The mixture was concentrated in vacuo and the residue was suspended in aqueous hydrochloric acid (500 mL, 1 M solution) and stirred at RT for 18 hours. After this time the solid was collected by filtration and washed with water (x 3). The solid was partially dissolved in ethyl acetate and heated at reflux for 2 hours. The remaining solid was removed by hot filtration and the filtrate was evaporated to afford the title compound. The solid collected from the hot ethyl acetate was again partially dissolved in ethyl acetate and heated at reflux for 2 hours then filtered to give filtrate containing pure product. This process was repeated four more times. The combined solid was recrystallised from ethyl acetate and petroleum ether to afford the title compound (20.0 g, 76%). NMR (400 MHz, DMSO): delta 10.68 (s, 1 H), 8.19 (d, J = 9.9 Hz, 1 H), 7.58 (d, J = 7.5 Hz, 2 H), 7.41 -7.36 (m, 2 H), 7.34-7.29 (m, 1 H), 7.23-7.19 (m, 2 H), 6.57 (d, J = 9.8 Hz, 1 H), 5.94 (d, J = 4.7 Hz, 1 H), 5.31 (s, 2 H), 5.25-5.19 (m, 1 H), 3.71 -3.58 (m, 2 H). | |
| 76% | 8-(Benzyloxy)-5-(2-bromoacetyl)quinolin-2(lH)-one (26.069.9 mmol) and fR -S^-diphenyl- l-methyltetrahydro-SH-pyrrolof l ^- c][l ,3,2]oxazaborole (21.3 g, 76.8 mmol) were azeotroped with toluene (x 3) then suspended in anhydrous THF (400 mL) under an atmosphere of nitrogen. The suspension was cooled to -20C (external temperature) and borane dimethyl sulfide complex solution (45.4 mL, 90.8 mmol, 2.0 M solution in THF) was added by syringe pump over 3 hours. After complete addition the reaction mixture was stirred for one hour before quenching with methanol (25 mL). The reaction was warmed to RT over 20 minutes. The mixture was concentrated under reduced pressure and the residue was suspended in aqueous hydrochloric acid (500 mL, 1 M solution) and stirred at RT for 18 hours. After this time the solid was collected by filtration and washed with water (3 x 100 mL). The solid was partially dissolved in ethyl acetate and heated at reflux for 2 hours. The remaining solid was removed by hot filtration and the filtrate was evaporated to afford the title compound. The solid collected from the hot ethyl acetate was again partially dissolved in ethyl acetate and heated at reflux for 2 hours then filtered to give filtrate containing pure product. This process was repeated four more times. The combined solid was recrystallised from ethyl acetate and petroleum ether to afford the title compound (20.0 g, 76%). NMR (400 MHz, DMSO-d6): delta 10.68 (s, 1 H); 8.19 (d, J = 9.9 Hz, 1 H); 7.58 (d, J = 7.5 Hz, 2 H); 7.41 -7.36 (m, 2 H); 7.34-7.29 (m, 1 H); 7.23-7.19 (m, 2 H); 6.57 (d, J = 9.8 Hz, 1 H); 5.94 (d, J = 4.7 Hz, 1 H); 5.31 (s, 2 H); 5.25-5.19 (m, 1 H); 3.71 -3.58 (m, 2 H). | |
| 76% | 8-(Benzyloxy)-5-(2-bromoacetyl)quinolin-2(1H)-one (26.0 g, 69.9 mmol) and (R)-3,3-diphenyl-1-methyltetrahydro-3H-pyrrolo[1,2-c][1,3,2]oxazaborole (21.3 g, 76.8 mmol) were azeotroped with toluene (*3) then suspended in anhydrous THF (400 mL) under an atmosphere of nitrogen. The suspension was cooled to -20 C. (external temperature), and borane dimethyl sulfide complex solution (45.4 mL, 90.8 mmol, 2.0 M solution in THF) was added by syringe pump over 3 hours. After complete addition the reaction mixture was stirred for an hour before quenching with methanol (25 mL). The reaction was warmed to RT over 20 minutes. The mixture was concentrated in vacuo and the residue was suspended in aqueous hydrochloric acid (500 mL, 1 M solution) and stirred at RT for 18 hours. After this time, the solid was collected by filtration and washed with water (*3). The solid was partially dissolved in ethyl acetate and heated at reflux for 2 hours. The remaining solid was removed by hot filtration, and the filtrate was evaporated to afford the title compound. The solid collected from the hot ethyl acetate was again partially dissolved in ethyl acetate and heated at reflux for 2 hours then filtered to give filtrate containing pure product. This process was repeated four more times. The combined solid was recrystallised from ethyl acetate and petroleum ether to afford the title compound (20.0 g, 76%). 1H NMR (400 MHz, DMSO): delta 10.68 (s, 1H), 8.19 (d, J=9.9 Hz, 1H), 7.58 (d, J=7.5 Hz, 2H), 7.41-7.36 (m, 2H), 7.34-7.29 (m, 1H), 7.23-7.19 (m, 2H), 6.57 (d, J=9.8 Hz, 1H), 5.94 (d, J=4.7 Hz, 1H), 5.31 (s, 2H), 5.25-5.19 (m, 1H), 3.71-3.58 (m, 2H). | |
| 76% | With dimethylsulfide borane complex; (3aR)-1-methyl-3,3-diphenyl-tetrahydro-pyrrolo[1,2-c][1,3,2]oxazaborole; In tetrahydrofuran; at -20℃; for 4h; | Step 2: (R)-8-(Benzyloxy)-5-(2-bromo-1-hydroxyethyl)quinolin-2(1H)-one 8-(Benzyloxy)-5-(2-bromoacetyl)quinolin-2(1H)-one (26.0 g, 69.9 mmol) and (R)-3,3-diphenyl-1-methyltetrahydro-3H-pyrrolo[1,2-c][1,3,2]oxazaborole (21.3 g, 76.8 mmol) were azeotroped with toluene (*3) then suspended in anhydrous THF (400 mL) under an atmosphere of nitrogen. The suspension was cooled to -20 C. (external temperature) and borane dimethyl sulfide complex solution (45.4 mL, 90.8 mmol, 2.0 M solution in THF) was added by syringe pump over 3 hours. After complete addition the reaction mixture was stirred for one hour before quenching with methanol (25 mL). The reaction was warmed to RT over 20 minutes. The mixture was concentrated under reduced pressure and the residue was suspended in aqueous hydrochloric acid (500 mL, 1 M solution) and stirred at RT for 18 hours. After this time the solid was collected by filtration and washed with water (3*100 mL). The solid was partially dissolved in ethyl acetate and heated at reflux for 2 hours. The remaining solid was removed by hot filtration and the filtrate was evaporated to afford the title compound. The solid collected from the hot ethyl acetate was again partially dissolved in ethyl acetate and heated at reflux for 2 hours then filtered to give filtrate containing pure product. This process was repeated four more times. The combined solid was recrystallised from ethyl acetate and petroleum ether to afford the title compound (20.0 g, 76%). 1H NMR (400 MHz, DMSO-d6): delta 10.68 (s, 1H); 8.19 (d, J=9.9 Hz, 1H); 7.58 (d, J=7.5 Hz, 2H); 7.41-7.36 (m, 2H); 7.34-7.29 (m, 1H); 7.23-7.19 (m, 2H); 6.57 (d, J=9.8 Hz, 1H); 5.94 (d, J=4.7 Hz, 1H); 5.31 (s, 2H); 5.25-5.19 (m, 1H); 3.71-3.58 (m, 2H). |
| 76% | With dimethylsulfide borane complex; (3aR)-1-methyl-3,3-diphenyl-tetrahydro-pyrrolo[1,2-c][1,3,2]oxazaborole; In tetrahydrofuran; toluene; at -20 – 20℃;Inert atmosphere; | 8-(Benzyloxy)-5-(2-bromoacetyl)quinolin-2(lH)-one (26.0 g, 69.9 mmol) and (^-3,3-diphenyl- 1 -methyltetrahydro-3H-pyrrolo[ 1 ,2-c][ 1 ,3,2]oxazaborole (21.3 g, 76.8 mmol) were azeotroped with toluene (x 3) then suspended in anhydrous THF (400 mL) under an atmosphere of nitrogen. The suspension was cooled to -20 C (external temperature) and borane dimethyl sulfide (BH3-Me2S) complex solution (45.4 mL, 90.8 mmol, 2.0 M solution in THF) was added by syringe pump over 3 hours. After complete addition the reaction mixture was stirred for one hour before quenching with methanol (25 mL). The reaction was warmed to RT over 20 minutes. The mixture was concentrated under reduced pressure and the residue was suspended in aqueous hydrochloric acid (500 mL, 1 M solution) and stirred at RT for 18 hours. After this time the solid was collected by filtration and washed with water (3 x 100 mL). The solid was partially dissolved in ethyl acetate and refluxed for 2 hours. The remaining solid was removed by hot filtration and the filtrate was evaporated to afford the title compound. The solid collected from the hot ethyl acetate was again partially dissolved in ethyl acetate and refluxed for 2 hours then filtered to give filtrate containing pure product. This process was repeated four more times. The combined solid was recrystallised from ethyl acetate and petroleum ether to afford the title compound (20.0 g, 76%). 1HNMR (400 MHz, DMSO-d6): delta 10.68 (s, 1 H); 8.19 (d, J = 9.9 Hz, 1 H); 7.58 (d, J = 7.5 Hz, 2 H); 7.41-7.36 (m, 2 H); 7.34-7.29 (m, 1 H); 7.23-7.19 (m, 2 H); 6.57 (d, J = 9.8 Hz, 1 H); 5.94 (d, J = 4.7 Hz, 1 H); 5.31 (s, 2 H); 5.25-5.19 (m, 1 H); 3.71-3.58 (m, 2 H). |
| 76% | 8-(l3enzyloxy)-5-(2-bromoacetyl)quinolin-2(1 H)- one (26.0 g, 69.9 mmol) and (R)-3,3-diphenyl-1-methyltet- rahydro-3H-pyrrolo[1 ,2-c] [1 ,3,2]oxazaborole (21.3 g, 76.8 mmol) were azeotroped with toluene (x3) then suspended in anhydrous THF (400 mE) under an atmosphere of nitrogen. The suspension was cooled to -20 C. (external temperature) and borane dimethyl sulfide complex solution (45.4 mE, 90.8 mmol, 2.0 M solution in THF) was added by syringe pump over 3 hours. Afier complete addition the reaction mixture was stirred for one hour before quenching with methanol (25 mE). The reaction was warmed to room temperature over 20 minutes. The mixture was concentrated under reduced pressure and the residue was suspended in aqueous hydrochloric acid (500 mE, 1 M solution) and stirred at room temperature for 18 hours. After this time the solid was collected by filtration and washed with water (3×100 mE). The solid was partially dissolved in ethyl acetate and heated at reflux for 2 hours. The remaining solid was removed by hot filtration and the filtrate was evaporated to afford the title compound. The solid collected from the hot ethyl acetate was again partially dissolved in ethyl acetate and heated at reflux for 2 hours then filtered to give filtrate containing pure product. This process was repeated four more times. The combined solid was recrystallised from ethyl acetate and petroleum ether to afford the title compound (20.0 g, 76%). 1H NMR (400 MHz, DMSO-d5): oe 10.68 (s, 1H),8.19 (d, J=9.9 Hz, 1H), 7.58 (d, J=7.5 Hz, 2H), 7.41-7.36 (m,2H), 7.34-7.29 (m, 1H), 7.23-7.19 (m, 2H), 6.57 (d, J=9.8 Hz,1H), 5.94 (d, J=4.7 Hz, 1H), 5.31 (s, 2H); 5.25-5.19 (m, 1H),3.71-3.58 (m, 2H). | |
| 76% | With borane dimethyl sulfide complex; (3aR)-1-methyl-3,3-diphenyl-tetrahydro-pyrrolo[1,2-c][1,3,2]oxazaborole; In tetrahydrofuran; toluene; at -20℃; for 4h;Inert atmosphere; | 8-(benzyloxy)-5-(2-bromoacetyl)quinolin-2(1H)-one (26.0 g, 69.9 mmol) and (R)- 3 ,3-diphenyl- 1 -methyltetrahydro-3H-pyrrolo [ 1 ,2-c] [ 1 ,3 ,2]oxazaborole (21.3 g, 76.8 mmol) were azeotroped with toluene (x 3) then suspended in anhydrous THF (400 mL) under an atmosphere of nitrogen. The suspension was cooled to -20C (external temperature) and borane dimethyl sulfide complex solution (45.4 mL, 90.8 mmol, 2.0 M solution in THF) was added by syringe pump over 3 hours. After complete addition the reaction mixture was stirred for one hour before quenching with methanol (25 mL). The reaction was warmed to room temperature over 20 minutes. The mixture was concentrated under reduced pressure and the residue was suspended in aqueous hydrochloric acid (500 mL, 1 M solution) and stirred at room temperature for 18 hours. After this time the solid was collected by filtration and washed with water (3 x 100 mL). The solid was partially dissolved in ethyl acetate and heated at reflux for 2 hours. The remaining solid was removed by hot filtration and the filtrate was evaporated to afford the title compound. The solid collected from the hot ethyl acetate was again partially dissolved in ethyl aceta
Details |





Reviews
There are no reviews yet.